 Neither of the component amino acids is sweet (Phe is actually bitter), and derivatives of the other dipeptide (Phe-Asp) are not sweet.Ī tripeptide composed of three different amino acids can be made in 6 different constitutions, and the tetrapeptide shown above (composed of four different amino acids) would have 24 constitutional isomers. The methyl ester of the first dipeptide (structure on the right) is the artificial sweetener aspartame, which is nearly 200 times sweeter than sucrose. Thus, aspartic acid (Asp) and phenylalanine (Phe) may be combined to make Asp-Phe or Phe-Asp, remember that the amino acid on the left is the N-terminus. For example, a dipeptide made from two different amino acids may have two different structures. This aspect of peptide structure is an important factor influencing the conformations adopted by proteins and large peptides.īecause the N-terminus of a peptide chain is distinct from the C-terminus, a small peptide composed of different aminoacids may have a several constitutional isomers. The color shaded rectangles in the lower structure define these regions, and identify the relatively facile rotations that may take place where the corners meet (i.e. This keeps the peptide links relatively planar and resistant to conformational change. As shown in the following diagram, nitrogen electron pair delocalization into the carbonyl group results in significant double bond character between the carbonyl carbon and the nitrogen. 
This restriction is due to the rigid nature of the amide (peptide) bond. The conformational flexibility of peptide chains is limited chiefly to rotations about the bonds leading to the alpha-carbon atoms. The " Show Structure" button displays some bond angles and lengths that are characteristic of these compounds. As expected, the free amine and carboxylic acid functions on a peptide chain form a zwitterionic structure at their isoelectric pH.īy clicking the " Grow Peptide" button, an animation showing the assembly of this peptide will be displayed. By convention, the amino acid component retaining a free amine group is drawn at the left end (the N-terminus) of the peptide chain, and the amino acid retaining a free carboxylic acid is drawn on the right (the C-terminus). A simple tetrapeptide structure is shown in the following diagram. If the amine and carboxylic acid functional groups in amino acids join together to form amide bonds, a chain of amino acid units, called a peptide, is formed. It is extremely important to understand the physical forces behind a peptide bond, as this allows scientists to design accurate, predictive models of three-dimensional protein structures.Proteins Peptides & Proteins 1. A mistake in the translation process can lead to protein mis-folding, and in turn, disease. Proteins can be as small as forty-four amino acids, or as large as thirty-five thousand. 
Peptide bonds are made within ribosomes during a process called «translation» to form polypeptides, which then undergo various molecular processing and modification, before folding into a three-dimensional shape, which we call a protein. For instance, there is currently much interest in antibody-drug conjugates these pair fragmented antibodies with pharmacologically active compounds in order to specifically target cancer tumors, among other things. The ability to predictably split peptide bonds is vital to a number of different fields of study. Reversing a peptide bond without an enzyme is extremely difficult, thus this process is usually mediated by an enzyme called a protease, such as subtilisin, which is frequently added to laundry detergent to cleave many protein contaminants. It may therefore be counterintuitive to learn that peptide bonds are quite stable kinetically: the lifetime of a peptide bond in aqueous solution is approximately 1000 years. Hence, the biosynthesis of a peptide bond requires an input of free energy. One interesting thing to note is that the equilibrium of this reaction lies on the side of hydrolysis rather than synthesis. For most peptides the cis-form is about 1000 times less stable than the trans-form. In such cases, the cis form is more stable than usual since the proline side-chain offers less of a hindrance. However, cis forms can occur in peptide bonds that precede a proline residue. 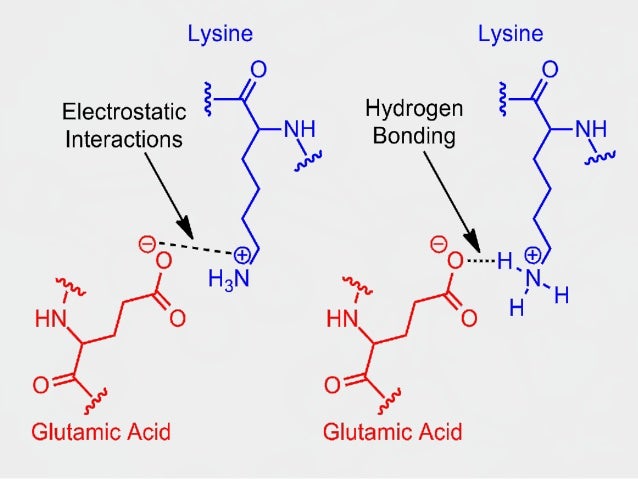
In naturally occurring peptides most peptide bonds are in the trans configuration. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
